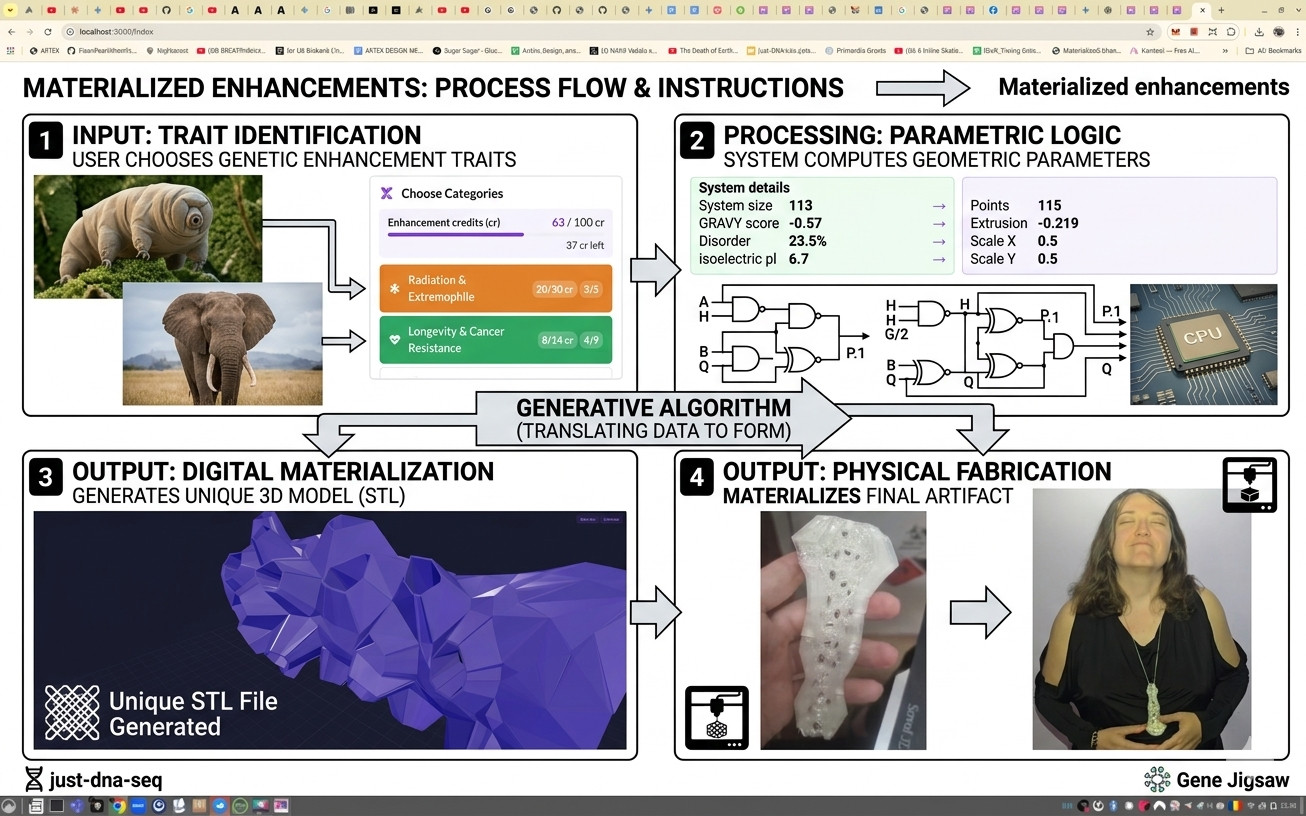
Stress Resistance0/8 genes selected · 0 crExpand
Dsup is a tardigrade protein that can help shield DNA from some forms of radiation damage, acting a bit like a protective wrap around chromosomes. Early mouse and human-cell work is promising, but it also behaved badly in nerve cells, so this is not a simple universal shield.
Bowhead whales seem to use CIRBP as part of a very careful DNA-repair system that helps cells fix breaks cleanly. Human-cell and fly experiments look encouraging, but nobody has yet shown that it makes mammals live longer or resist cancer safely.
SOD2 helps mitochondria clean up reactive oxygen stress, which sounds like it should slow aging. In long-running mouse studies, extra SOD2 reduced some damage but did not extend lifespan, making it more of a survival helper than a longevity switch.
CAHS D is a tardigrade protein that helps cells cope with drying-like stress by forming reversible protective structures. It works modestly in human cells under lab stress, but true mammalian drying survival remains far beyond current evidence.
Deinococcus uses PprI and related repair systems to survive radiation levels that would destroy most life. A few mouse and human-cell studies suggest PprI can reduce radiation damage, but the result needs independent replication and safer delivery ideas.
Melanized fungi from extreme environments can absorb radiation and neutralize damaging chemistry, which may explain part of their toughness. The more exotic idea that they harvest radiation like food is still debated, and melanin can also make tumors harder to treat.
Weddell seals store huge amounts of oxygen in muscle using myoglobin that stays soluble at extreme concentrations. The diving phenotype depends on whole-body physiology, so seal myoglobin alone is not yet a proven upgrade for mammalian cells.
Antifreeze proteins from polar fish stick to tiny ice crystals and slow their growth, which is useful for preserving cells and organs. They are promising for cryobiology, but chronic use in living bodies is unproven and too much can damage tissue.
LEA proteins from the sleeping chironomid form glassy protective matrices during desiccation, a distinct mechanism from tardigrade CAHS gels. They have been expressed in mammalian cells and protect against drying damage, with strongest relevance to dry biopreservation.
The sleeping chironomid expanded its protein-repair enzyme PIMT to 14 copies, enabling aggressive repair of age-damaged amino acid residues during desiccation and radiation stress. Humans have one copy; boosting this pathway could enhance protein quality control.
TDR1 is a tardigrade-specific nuclear protein that helps cells survive DNA-damaging agents, working through damage response rather than shielding like Dsup. It improved human cell survival against radiomimetic drugs, suggesting tardigrades have a layered radioprotection system.
The sleeping chironomid repurposed its HSF1 heat-shock transcription factor to activate an entire desiccation survival program. HSF1 knockdown cuts survival fivefold. The principle — rewiring a conserved human transcription factor to drive broader protection — is an engineering blueprint, not a gene transfer.
Trehalose is the universal desiccation protectant in nature — the sleeping chironomid accumulates it to 20% dry weight before drying out. Mammalian cells lack the gene entirely, but engineered trehalose synthesis has improved human cell survival during drying, making this a key target for biopreservation.
Marsupials retain a CPD photolyase gene that placental mammals lost 170 million years ago. Restoring it via mRNA delivery to human skin cells enables light-activated DNA repair that dramatically reduces UV damage and skin cancer risk.
Longevity & Genome0/11 genes selected · 0 crExpand
Naked mole-rats make an unusually large, springy form of hyaluronic acid, and their version of HAS2 helped mice live a little longer with fewer cancers. The effect is real but modest, and the same biology can behave differently in tumor settings.
FOXO3 is one of the strongest human genetic clues linked to exceptional old age, especially in centenarian studies. It turns on repair and stress-response programs, but the key gain-of-function experiment in mammals is still missing.
Klotho is a hormone-like protein tied to healthier aging, better cognition, and longer life in some mouse studies. It has a narrow sweet spot: too little is harmful, but too much or the wrong variant can also cause problems.
Elephants carry many extra TP53 copies, giving their cells a stronger tendency to self-destruct when DNA damage looks dangerous. That may help explain their low cancer burden, but turning up TP53 in humans could also accelerate aging or harm healthy tissue.
MGMT-P140K is an engineered DNA-repair variant that has already helped protect patient bone marrow during chemotherapy trials. It is one of the most clinically grounded genes here, but it protects against a narrow kind of damage and can be risky in the wrong cells.
Elephant p53-R9 is a stripped-down TP53 retrogene that can push damaged cancer cells into self-destruction through mitochondria. It is exciting as a cancer-cell killing mechanism, but it has not yet been tested in normal tissues or living animals.
Greenland sharks live for centuries, and their genome hints at expanded DNA-repair machinery that may support that longevity. For now this is mostly a map of clues, not a tested enhancement, because the shark genes have barely been functionally studied.
A single base edit that permanently knocks out PCSK9 in the liver, lowering LDL cholesterol for life without daily pills. Monkey studies showed 59% LDL reduction lasting over a year, and early human trials confirm durable effects.
APOE4 is the strongest genetic risk factor for Alzheimer's, and rare protective variants like APOE2 and the Christchurch mutation dramatically reduce disease. Gene editing to convert a patient's APOE4 to a protective form is compelling but has never been achieved in a living brain.
FGF21 and a soluble TGF-beta trap were combined in a single gene therapy that reversed obesity, diabetes, heart failure, and kidney fibrosis in mouse disease models. FGF21 alone extends mouse lifespan 36%. Whether the combination extends lifespan in naturally aging animals is still unknown.
The sleeping chironomid expanded its protein-repair enzyme PIMT to 14 copies, enabling aggressive repair of age-damaged amino acid residues during desiccation and radiation stress. Humans have one copy; boosting this pathway could enhance protein quality control.
TDR1 is a tardigrade-specific nuclear protein that helps cells survive DNA-damaging agents, working through damage response rather than shielding like Dsup. It improved human cell survival against radiomimetic drugs, suggesting tardigrades have a layered radioprotection system.
Chlorotoxin from the deathstalker scorpion selectively binds brain tumors and has completed Phase I/II human clinical trials for glioma imaging and targeted radiotherapy — one of the most successful venom-to-medicine translations.
RBM3 is a cold-shock protein that explains why hibernating animals' brains survive near-freezing temperatures. Overexpression alone — without cooling — prevented neurodegeneration and extended survival in Alzheimer's and prion mouse models.
Environmental Adaptation0/12 genes selected · 0 crExpand
Bowhead whales seem to use CIRBP as part of a very careful DNA-repair system that helps cells fix breaks cleanly. Human-cell and fly experiments look encouraging, but nobody has yet shown that it makes mammals live longer or resist cancer safely.
AQP1 is a water-channel protein that helps cells move water quickly, and mammalian AQP1 biology is well understood. The famous water-holding frog story is much less proven: its special AQP1 has not actually been cloned and tested as an enhancement.
The Tibetan EPAS1 variant helps people live at high altitude without over-thickening their blood. It is a beautiful human evolution story, but nearby changes in the same oxygen-sensing pathway can also drive dangerous blood and tumor disorders.
Bats carry a softened version of STING that may help them avoid the runaway inflammation that makes many viral infections deadly. A bat-like mouse variant reduced some age-linked inflammation, but it may also weaken tumor surveillance or antiviral defenses.
Weddell seals store huge amounts of oxygen in muscle using myoglobin that stays soluble at extreme concentrations. The diving phenotype depends on whole-body physiology, so seal myoglobin alone is not yet a proven upgrade for mammalian cells.
Antifreeze proteins from polar fish stick to tiny ice crystals and slow their growth, which is useful for preserving cells and organs. They are promising for cryobiology, but chronic use in living bodies is unproven and too much can damage tissue.
Dolphin-like clock-gene changes can make zebrafish larvae sleep less and stay more wakeful in experiments. That is a real hint about sleep biology, but dolphin half-brain sleep is a brain-network feat, not something a single gene can install.
Electric fish repurposed sodium-channel genes like scn4aa as part of organs that fire thousands of cells in series. The channel biology is fascinating, but the impressive voltage comes from anatomy, not from one protein acting alone.
Trehalose is the universal desiccation protectant in nature — the sleeping chironomid accumulates it to 20% dry weight before drying out. Mammalian cells lack the gene entirely, but engineered trehalose synthesis has improved human cell survival during drying, making this a key target for biopreservation.
Marsupials retain a CPD photolyase gene that placental mammals lost 170 million years ago. Restoring it via mRNA delivery to human skin cells enables light-activated DNA repair that dramatically reduces UV damage and skin cancer risk.
Mongooses resist cobra venom through a single amino acid change (F187N) in the nicotinic acetylcholine receptor that blocks neurotoxin binding without affecting normal nerve signaling — convergently evolved at least 13 times across mammals.
RBM3 is a cold-shock protein that explains why hibernating animals' brains survive near-freezing temperatures. Overexpression alone — without cooling — prevented neurodegeneration and extended survival in Alzheimer's and prion mouse models.
Regeneration0/8 genes selected · 0 crExpand
TERT rebuilds telomeres, the protective caps that shorten as cells divide, and mouse gene therapy studies have extended lifespan without extra cancer in those experiments. The danger is that many human cancers reactivate TERT, so safe use would require very careful control.
The immortal jellyfish can reverse its life cycle, and its telomere-related genes are tempting suspects in that age-resetting trick. The tested POT1 change only weakened DNA binding in a lab assay, and similar changes in humans can raise cancer risk.
Planarian smedwi genes help maintain stem cells that let flatworms regrow whole bodies from tiny fragments. Mammals use related PIWI genes mostly in germ cells, and when similar programs switch on in body tissues they often look more like cancer than regeneration.
Lin28a can push cells toward a younger, growth-ready state and helped young mice heal ear and digit injuries faster. Its regenerative boost fades in adult tissues, and the same pathway can loosen restraints on cancer-linked genes.
Follistatin-344 neutralises the muscle-growth brake myostatin. AAV gene therapy trials in muscular dystrophy patients showed safety and modest functional improvement, but follistatin's broad activity across TGF-beta family members makes long-term effects unpredictable.
Myostatin is the body's brake on muscle growth. Natural knockouts in cattle, dogs, and one human child show dramatic muscle hypertrophy with few health consequences, making MSTN disruption a compelling target for treating muscle-wasting diseases.
VEGF gene therapy aims to grow new blood vessels in starved heart or limb tissue by injecting DNA encoding a vascular growth factor. Early trials showed promise, but larger placebo-controlled studies have not confirmed lasting clinical benefit.
Spiny mice are the only mammals that regenerate full-thickness skin without scarring. Their wounds heal with hair follicles and glands restored, aided by unique hemostasis and reduced fibrotic signaling. The molecular program is still being mapped — a systems-level adaptation, not a single gene.
Perception0/8 genes selected · 0 crExpand
Prestin is the motor protein that helps inner-ear cells amplify sound, and echolocating bats and dolphins carry strikingly similar versions of it. The protein changes are measurable in cells, but nobody has used them to give a mammal ultrasonic hearing.
Pit-viper TRPA1 is a heat-sensitive channel that can detect warm prey through infrared-like thermal cues. The channel works in lab cells, but true heat vision also needs the specialized pit organ anatomy that mammals do not have.
Robin CRY4a is a light-sensitive protein that may help migratory birds sense Earths magnetic field through quantum-scale chemistry. The purified protein is magnetically responsive in the lab, but recreating a compass in mammalian cells looks biochemically difficult.
The cat tapetum is a mirror-like eye layer that bounces dim light back through the retina for a second chance at detection. It is an engineered tissue architecture rather than a single gene, so cat-style night vision is not a straightforward genetic add-on.
Birds see UV light through a version of the blue cone opsin (SWS1) that differs from the human version by just 2–3 amino acids near the chromophore. Retuning human OPN1SW to UV sensitivity is molecularly simple but optically complicated by the UV-blocking lens.
PIEZO2 is the Nobel Prize-winning mechanosensitive ion channel responsible for light touch and proprioception. Increasing its density in skin could create superhuman tactile sensitivity, but gain-of-function mutations elsewhere cause joint disorders.
TMC1 is the pore-forming channel that converts sound into nerve signals in the inner ear. AAV gene therapy delivering TMC1 to deaf mice fully restores hearing, and clinical translation is underway.
Sharks sense electric fields through a specialized splice variant of the BK potassium channel that lacks the STREX exon. Expressing this variant in human skin cells could theoretically create a new sense, but no mammal has the neural circuitry for it.
Expression0/8 genes selected · 0 crExpand
Cephalopod reflectins can reorganize to change how cells scatter light, helping squid and cuttlefish tune their shimmering skin. Human-cell experiments show controllable optical changes, but real camouflage would also require specialized skin organs and neural control.
GFP is the jellyfish protein that made living cells glow green and transformed biology as a lab tool. It works beautifully as a reporter across many organisms, but visible fluorescence does not penetrate human tissue well and is not a therapy.
Firefly luciferase makes light by burning luciferin, letting researchers watch cells inside living animals after giving the substrate. It is powerful for imaging, but a self-glowing mammal still faces the hard limits of fuel supply and light blocked by tissue.
Gecko beta-keratin proteins help build the microscopic hairs that let gecko feet stick by van der Waals forces. The stickiness comes from a precise hierarchy of skin structures, so expressing the protein alone would not grow a gecko toe pad.
Spider dragline silk protein MaSp1 produces fibers stronger than steel and more elastic than nylon. It has been expressed in transgenic goats, silkworms, and sheep embryos, but full-length production remains a challenge.
Chlorotoxin from the deathstalker scorpion selectively binds brain tumors and has completed Phase I/II human clinical trials for glioma imaging and targeted radiotherapy — one of the most successful venom-to-medicine translations.
Resilin is an insect elastomeric protein that returns 92–97% of stored energy — the most efficient biological rubber known. Recombinant resilin is being developed for tissue engineering of tendons, vocal cords, and cardiovascular grafts.
Pif orchestrates nacre (mother-of-pearl) formation in oysters, creating a nanostructured composite 3,000× tougher than pure mineral. Its two domains anchor to scaffold and nucleate crystals — a template for biomimetic bone reinforcement.
How it works